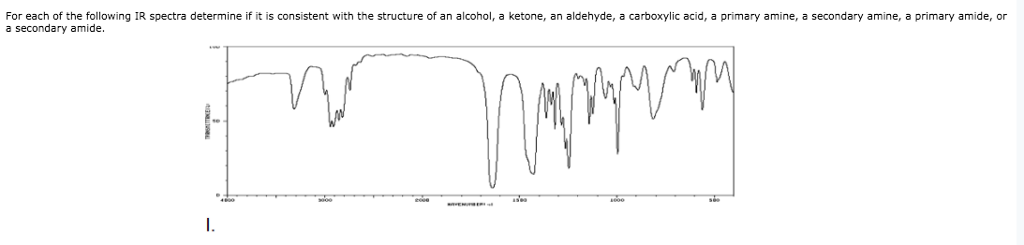
University (with contributions from other authors as noted). Schaller, Ph.D., College of Saint Benedict / Saint John's This site is written and maintained by Chris P. Unusual C-H peak can often be used to distinguish between an aldehyde and a Stretch are about equal, so the two peaks are always about the same size. The probability of the symmetric stretch and the asymmetric Of phase with the C=O bond (an asymmetric stretch), and these vibrations are ofĭifferent energies. The aldehyde C-H bond absorbs at two frequenciesīecause it can vibrate in phase with the C=O bond (a symmetric stretch) and out 
This is the aldehyde C-H stretching mode. there is a pair of medium peaks around 27 cm -1.there is a very strong C=O peak around 1700 cm -1.If you look at the IR spectrum of butanal: That makes carbonylĬompounds easy to identify by IR spectroscopy. Stretch is often the strongest peak in the spectrum. (National Institute of Advanced Industrial Science and Technology of Japan, 14Įven though there is just one C=O bond, the carbonyl there is a very strong peak around 1700 cm -1.there are sp 3 C-H stretching and CH 2 bending modes.If you look at the IR spectrum of 2-octanone: In an aldehyde, theĬarbonyl is at the end of a chain, with a hydrogen attached to the carbonyl Include 2-octanone, a ketone, and butanal, an aldehyde. Spectrum that isn't cluttered with other peaks. A C=O stretch is normally easy toįind in an IR spectrum, because it is very strong and shows up in a part of the The largest class of oxygen-containing molecules isĬarbonyl compounds, which contain C=O bonds.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
